 
This is because as the number of atoms in a molecule increases, the probability that the molecule contains at least one heavy isotope atom also increases. The mass spectrum of Fe represents that the monoisotopic mass is not always the most abundant isotopic peak in a spectrum despite it containing the most abundant isotope for each atom. If a piece of iron was put into a mass spectrometer to be analyzed, the mass spectra of iron (Fe) would result in multiple mass spectral peaks due to the existence of the iron isotopes, 54 For example, when comparing tyrosine which has a molecular structure of C 9H 11NO 3 with a monoisotopic mass of 182.081 Da and methionine sulphone C 5H 11NO 4S which clearly are 2 different compounds but methionine sulphone has a 182.048 Da. The monoisotopic mass is very useful when analyzing small organic compounds since compounds with similar weights will not be differentiated if the nominal mass is used. Note that the masses used are neither the integer mass numbers nor the terrestrially averaged standard atomic weights as found in a periodic table. Where it will be clear that two different molecules are going through the mass spectrometer. 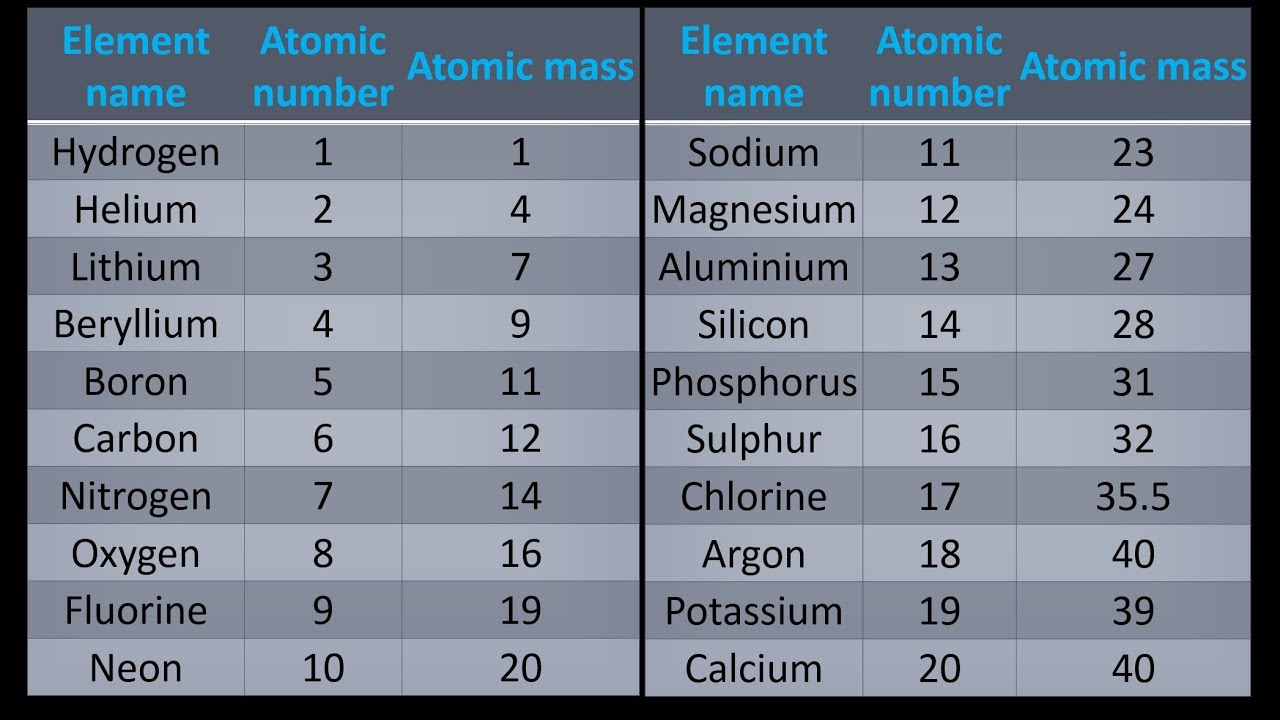
When calculating the monoisotopic masses, using the mass of the primary isotope of the elements including the mass defect: If a high-resolution instrument like an orbitrap or an ion cyclotron resonance is used, these two molecules can be distinguished. What this means, is when using mass spectrometer with insufficient source of power "low resolution" like a quadrupole mass analyser or a quadrupolar ion trap, these two molecules won't be able to be distinguished after ionization, this will be shown by the cross lapping of the m/z peaks. For example, when calculating the nominal mass of a molecule of nitrogen (N 2) and ethylene (C 2H 4) it comes out as. Nominal mass is a term used in high level mass spectrometric discussions, it can be calculated using the mass number of the most abundant isotope of each atom, without regard for the mass defect. Nominal mass vs monoisotopic mass Orbitrap Mass Analyzers Nominal mass 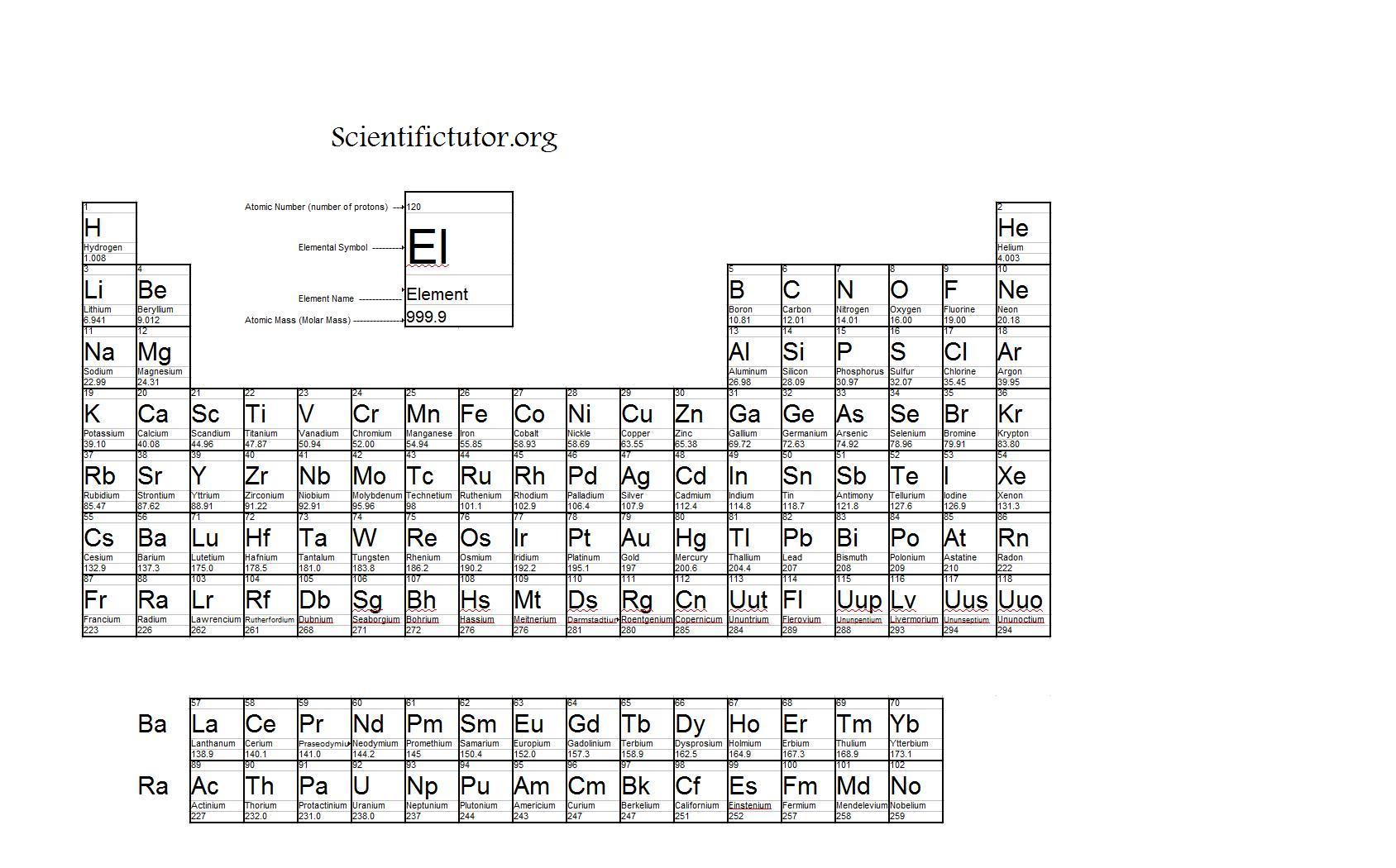
Monoisotopic mass is typically expressed in daltons (Da), also called unified atomic mass units (u). Iron's most common isotope has a mass number of 56, while the stable isotopes of iron vary in mass number from 54 to 58. However, this does not hold true for all atoms. For some atoms like carbon, oxygen, hydrogen, nitrogen, and sulfur, the M mi of these elements is exactly the same as the mass of its natural isotope, which is the lightest one. It also is different from the molar mass, which is a type of average mass. This differs from the nominal molecular mass, which is the sum of the mass number of the primary isotope of each atom in the molecule and is an integer. For small molecules made up of low atomic number elements the monoisotopic mass is observable as an isotopically pure peak in a mass spectrum. The theoretical monoisotopic mass of a molecule is computed by taking the sum of the accurate masses (including mass defect) of the most abundant naturally occurring stable isotope of each atom in the molecule. Monoisotopic mass (M mi) is one of several types of molecular masses used in mass spectrometry. For the elements with only one stable isotope, see monoisotopic element. This article is about the mass spectrometry term. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
